America
After Cough Syrup, Now Indian-Made Eye Drops Cause Concern; CDC Flags EzriCare
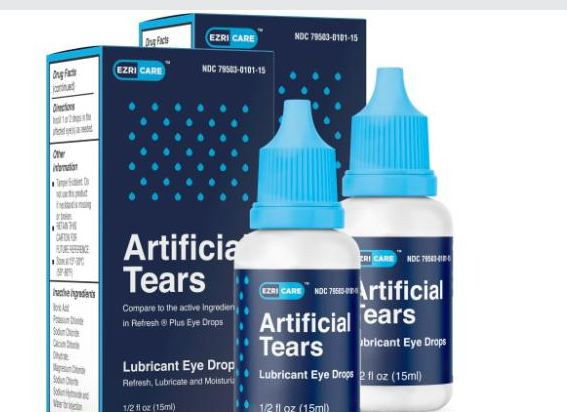
ATLANTA, GA: The US Center for Disease Control and Prevention (CDC) has flagged concern over the use of an Indian eye drop that is causing death and blindness.
The CDC has, in recent months, traced three deaths, eight cases of blindness and dozens of infections caused by a highly drug-resistant bacteria linked to EzriCare artificial tears, made by Global Pharma Healthcare, The New York Times reported.
Drug-resistant bacteria Pseudomonas aeruginosa has been a cause of concern among people with weak immune systems, residents of nursing homes and patients using catheters and breathing tubes.
In February, Global Pharma Healthcare recalled and stopped the production of both EzriCare Artificial Tears and Delsam Pharma’s Artificial Tears. And the US Food and Drug Administration (FDA) had instructed doctors and consumers not to purchase the product from the market and warned those who have already purchased it not to use it.
The warning issued by the FDA read, “using contaminated artificial tears increases the risk of eye infections that could result in blindness and deathâ€.
However, the CDC said it was concerned that the bacteria could gain a foothold in the US, as the bacteria showed signs of spreading among asymptomatic patients, in a Connecticut care center, who had the bacteria colonized in their bodies. Such spread tends to occur when patients touch common items or when healthcare workers transmit the germs.
According to experts, pseudomonas is very hard to get rid of. Pseudomonas is especially difficult to eradicate, both from health care facilities, where it clings tenaciously to sink drains, water faucets and other moist environments, and from patients who develop bloodstream infectionsâ€, David van Duin, an infectious disease specialist at the University of North Carolina School of Medicine, was quoted as saying.
After the FDA had issued the warning in February, a group of drug inspectors from the Central and Tamil Nadu governments conducted an inspection at the company premises which is 40 km south of Chennai.
While refusing to comment on the FDA’s remarks on the eyedrops, the Tamil Nadu drug regulator now said it found “no contamination†in samples of eye drops manufactured by Global Pharma, media reports said.
This is the third such incident where an Indian pharmaceutical product has come under scrutiny.
Last year, dozens of deaths were reported among children in Gambia and Uzbekistan due to Indian cough syrups.
Cough and cold syrups manufactured by New Delhi-based Maiden Pharmaceuticals were linked to the deaths of at least 70 children in Gambia, while Delhi-based Marion Biotech’s cough syrups were linked to the deaths of 18 children in Uzbekistan.

13 hours ago
Malavika Mohanan opens up on what recently made her fall in love with Mumbai a little more!

13 hours ago
Kajol celebrates husband, Ajay Devgn’s birthday with a funny twist

15 hours ago
NASA astronauts carry iPhones to Moon for 1st time aboard Artemis II mission

16 hours ago
US lifts sanctions on Venezuela's acting president

17 hours ago
RS: Oppn accuses govt of rushing through key bills for political 'gains' ahead of Assembly polls

17 hours ago
Not convinced that elections in India are free and fair, says Indian Overseas Congress chief Pitroda

17 hours ago
Priyanka Gandhi opens Kerala poll campaign slamming Centre and Left

17 hours ago
Tovino Thomas on how a church set for 'Pallichattambi' was so good that an old person thought it to be real!

17 hours ago
Akshay Kumar-starrer ‘Bhooth Bangla’ moves release date, to bow in cinemas on April 16

17 hours ago
Shilpa Shetty digs in nostalgia, shares what she was doing in the 90s

17 hours ago
Ranbir Kapoor’s no-show at ‘first look of Rama’ launch raises eyebrows

17 hours ago
Raj Kundra’s ‘The Great Punjab Robbery’ is locked for August 7 release

17 hours ago
Sanjay Dutt’s ‘Aakhri Sawaal’ teaser dives into fiery academic face-off, hidden past




















